Time-of-flight (TOF) is a simple but powerful technique that consists of accurately measuring the time it takes a particle/ atom/ ion/ molecule/ neutrino/ etc. to travel a known distance. This valuable tool has been used to characterise the kinetic energy distributions of an exhaustive range of sources, including positronium (Ps) [e.g. Howell et al, 1987], and is exploited widely in ion mass spectrometry.
Last year we published an article in which we described TOF measurements of ground-state (n=1) Ps atoms that were produced by implanting a short (5 ns) pulse of positrons into a porous silica film. Using pulsed lasers to photoionise (tear apart) the atoms at a range of well-defined positions, we were able to estimate the Ps velocity distribution, finding mean speeds on the order of 100 km/s. Extrapolating the measured flight paths back to the film’s surface indicated that the Ps took on average between 1 and 10 ns to escape the pores, depending on the depth to which the positrons were initially implanted.
When in the ground state and isolated in vacuum the electron and positron that make up a positronium atom will tend to annihilate each another in around 140 ns. Even with a speed of 100 km/s this means that Ps is unlikely to travel further than a couple of cm during its brief existence. Consequently, the photoionisation/ TOF measurements mentioned above were made within 6 mm of the silica film. However, instead of ionising the atoms, our lasers can be reconfigured to excite Ps to high-n Rydberg levels, and these typically live for a great deal longer. The increase in lifetime allows us to measure TOF spectra over much longer timescales (~10 µs) and distances (1.2 m).

The image above depicts the layout of our TOF apparatus. Positrons from a Surko trap are guided by magnets to the silica film, wherein they bind to electrons and are remitted as Ps. Immediately after, ultraviolet and infra-red pulsed lasers drive the atoms to n=2 and then to Rydberg states. Unlike the positively charged positrons, the neutral Ps atoms are not deflected by the curved magnetic fields and are able to travel straight along the 1.2 m flight tube, eventually crashing into the end of the vacuum chamber. The annihilation gamma rays are there detected using an NaI scintillator and photomultipler tube (PMT), and the time delay between Ps production and gamma ray detection is digitally recorded.
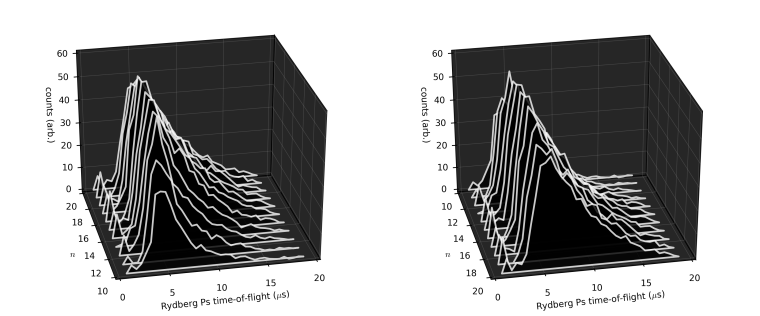
The plots above show two different views of time-of-flight spectra accumulated with the infra-red laser tuned to address Rydberg levels in the range of n=10 to 20. The data shows that more Ps are detected at later times for the higher-n states than for lower-n states. This is easily explained by fluorescence, i.e., the decay of an excited-state atom via spontaneous emission of a photon. As the fluorescence lifetime increases with n, the lower-n states are more likely to decay to the ground state and then annihilate before reaching the end of the chamber, reducing the number of gamma rays seen by the NaI detector at later times. We estimate from this data that Ps atoms in n=10 fluoresce in about 3 µs, compared to roughly 30 µs for n=20.
This work brings us an important step closer to performing a positronium free-fall measurement. A flight path of at least ten meters will probably be required to observe gravitational deflection, so we still have some way to go.
This post is based on work discussed in our article:
Measurement of Rydberg positronium fluorescence lifetimes. A. Deller, A. M. Alonso, B. S. Cooper, S. D. Hogan, and D. B. Cassidy. Phys. Rev. A 93, 062513 (2016)DOI:10.1103/PhysRevA.93.062513.

One thought on “How long does Rydberg positronium live?”